Though researchers are rapidly filling journals with data about the endocannabinoid system, we still don’t understand some of the basics of how this fundamental and pervasive neural signaling system works — for instance, how do endocannabinoids travel in and out of cells to get where they need to go?
This has been difficult to figure out for a variety of reasons. The body uses all sorts of different ways of transporting neurotransmitters around the body, all of which have been documented in research on other neural systems and the body at large. Specific mechanisms in other systems provide for transport of fat-soluble molecules like cannabinoids, but the evidence to dictate a solid model for endocannabinoid transport is still just over the horizon.
The theories are all controversial about how 2-AG and AEA (anandamide), the two human endocannabinoids, get where they need to go in the brain and peripheral nervous system.
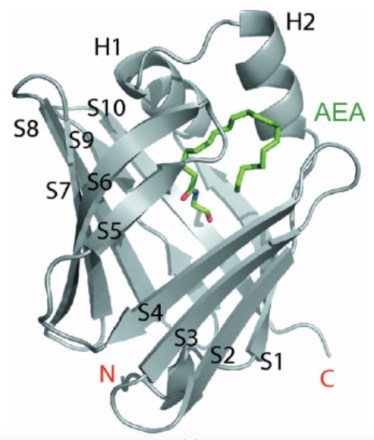
Some theories postulate specific molecules ferry endocannabinoids around inside and outside cells from synthesis to disassembly. These are called fatty acid binding proteins (FABPs), and there are several general-purpose ones that have been identified in other signaling systems. Some even show an ability to latch onto AEA and 2-AG, though that doesn’t necessarily prove they’re integral to the endocannabinoid system.
FABPs are numerous because the body uses all kinds of fat-based molecules for signaling and as components for larger functional molecules. They’re also important as targets for all sorts of pharmaceuticals that treat all sorts of conditions, considering the large role they play. As a result, geneticists have mapped the specific group of genes that code for known FABPs. The genes code for FABP 1 through 12, as well as other similar proteins, and share names with their proteins — i.e. FAPB5 is encoded by the FAPB5 gene, etc.
Some evidence that proteins known as ABCB1 and ABCG2 act as membrane transporters for cannabinoids, but they have not been definitively proven to be integral to the endocannabinoid system as of 2017 when the study mentioning them was published.
Another theory says really, AEA and 2-AG can get where they need to go by diffusion; that endocannabinoid concentrations inside cell structures drop as endocannabinoids break down, and as a result, suck more endocannabinoids through the membrane to replace the shortfall. This follows basic rules of fluid mechanics, and other body systems use the same principles to move molecules across membranes.
Still another theory involves “lipid rafts.” According to this model, the endocannabinoids travel by getting caught up in thicker concentrations of cholesterol and protein on cell membranes — the rafts. According to this theory, protein components in these relatively stationary, dense patches of cell membrane grapple onto the endocannabinoid and shove it into the CB (cannabinoid) receptors like CB1 and CB2.
Though still controversial, the most widely accepted theory of how anandamide and 2-AG move in and out of the cell is sort of a combination. Cellular uptake of endocannabinoids is governed by passive diffusion, but endocannabinoids get shuttled around within the cell by fatty acid binding proteins, specifically, FABP5, but maybe others, too.

Once the endocannabinoid molecule activates the receptor, it releases into the interior of the nerve cell where an FABP latches on and shuffles it to a little sack containing enzymes (FAAH and MAGL, discussed later) to break up the endocannabinoid into its components.
Once the endocannabinoids break apart, the concentration of endocannabinoid inside the cell drops lower than the concentration outside the cell, which causes the neuron to suck in more from the intercellular space, and the process repeats to maintain homeostasis.
Dale G. Deutsch of Stonybrook University was integral in early mapping of the endocannabinoid system — he was part of the team in 1993 who discovered FAAH, the enzyme that breaks down anandamide. Deutsch in a 2016 essay in Frontiers in Pharmacology outlined the endocannabinoid transport theory he spearheaded.
“Nearly all neurotransmitters are water-soluble and as such, require a transmembrane protein transporter to pass through the lipid membrane for inactivation inside the cell,” Deutch explains. “However, using model systems, we and others have shown that this is unnecessary for anandamide, an uncharged hydrophobic molecule that readily diffuses across the cellular membrane.
“Interestingly, its uptake is driven by the concentration gradient resulting from its breakdown mainly by FAAH localized in the endoplasmic reticulum,” Deutsch says. “We identified the FABPs as intracellular carriers that ‘solubilize’ anandamide, transporting anandamide to FAAH.”
He not only described how anadamide moves in and out of cells, but also how drugs could target FAPBs for therapeutic purposes.
“Compounds that bind to FABPs block AEA breakdown, raising its level,” Deutsch said. “The cannabinoids (THC and CBD) also were discovered to bind FABPs and this may be one of the mechanisms by which CBD works in childhood epilepsy, raising anandamide levels.”
Why don’t we know exactly how endocannabinoids travel and how they are transported?
Figuring out how endocannabinoids travel is difficult because researchers can’t watch all these interactions, even with the most powerful microscopes. As with all of the molecular interactions in pharmacology, researchers have to observe properties of the molecules indirectly. Even something as basic as the three-dimensional shape of a neurotransmitter or receptor has to be tortuously coaxed to reveal itself through crystallography or other indirect methods.
In this process, researchers have to find a molecule that will stick to the one they’re studying and also form a crystal, similar to your elementary school science class when you grew crystals with coal and Epsom salts. They can deduce the three-dimensional shape of the molecule by looking at the shape of the crystal it produces, because crystals by definition are an organized complex of repeating shapes that duplicate the shape of its smallest components. They figure out the shape of the crystal by bouncing X-rays off it and measuring the angles.
Scientists will also set up experiments with artificial membranes, dog guts, rat brains, synthetic enzymes, and dozens of different commercially produced inhibitors and agonist chemicals. They’ll tag chemicals with radioactive dye, or bombard them with ultraviolet light, ultimately to figure out exactly how each tooth of each gear of the endocannabinoid system fits with every other.

